Post by : Mariam Al-Faris
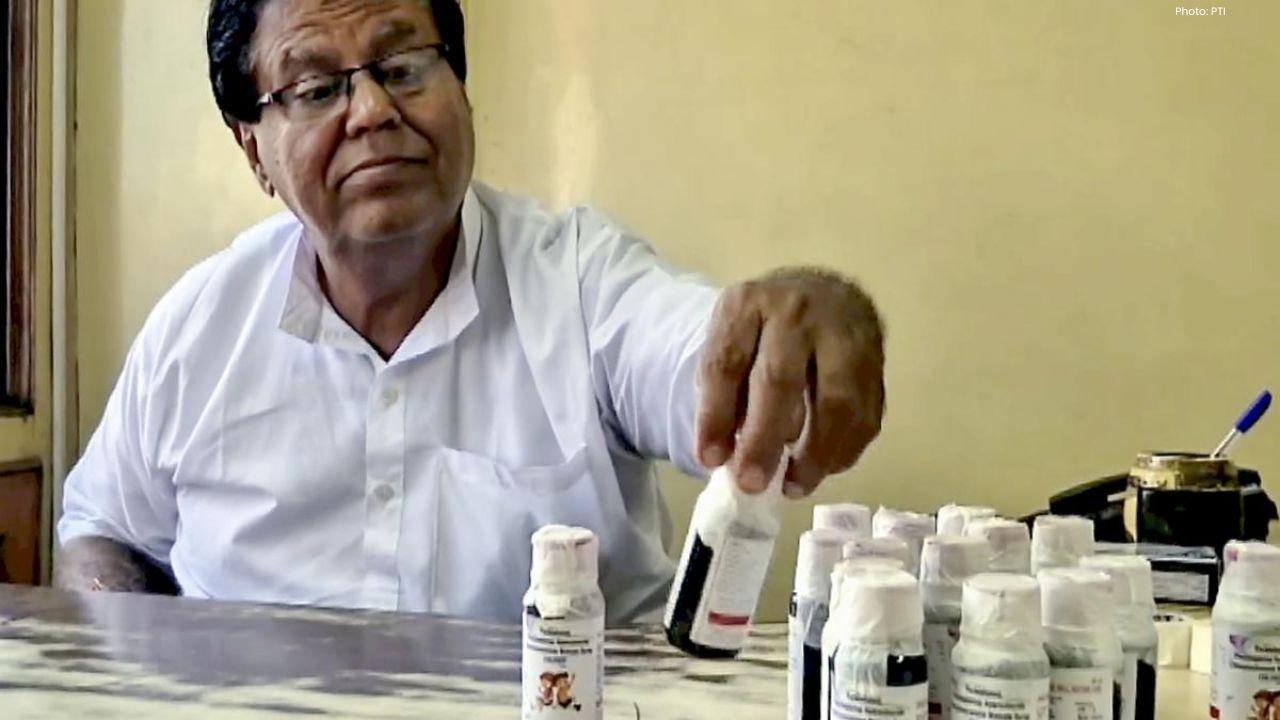
Ranganathan Govindan, the owner of Tamil Nadu-based Sresan Pharmaceuticals, has been arrested in connection with the adulterated Coldrif cough syrup that caused multiple children’s deaths across India. The Madhya Pradesh police took him into custody in Chennai late last night after extensive investigations into the scandal.
Deaths Linked to Poisonous Syrup
The case was filed after at least 20 children in Madhya Pradesh and several in Rajasthan died following the consumption of Coldrif syrup. The children suffered kidney infections caused by toxic substances in the cough syrup. Authorities charged Ranganathan with adulteration, culpable homicide not amounting to murder, and endangering the safety of children.
Meticulous Police Operation
Ranganathan had been on the run since the tragedy. Madhya Pradesh police, with the help of local Chennai authorities, launched a carefully planned midnight operation to arrest him. The team, which included drug inspectors, had been monitoring his movements and residence since October 5. He was caught around 1:30 am and taken to his company’s Kancheepuram factory, where crucial documents were seized.
Investigation to Widen
The police are now seeking a transit remand from a Chennai court to bring Ranganathan to Chhindwara, where most deaths occurred. Investigations will expand to cover the entire supply chain, including chemical suppliers, stockists, and medical representatives, to identify all parties responsible for distributing the toxic syrup.
Toxic Substance in Coldrif
Cold symptoms like runny nose, sneezing, sore throat, and watery eyes in children were treated using Coldrif. However, Tamil Nadu authorities found that the syrup contained diethylene glycol (DEG), a poisonous chemical used in inks and glue. DEG can severely damage kidneys, liver, and the nervous system. The factory was adding 46-48% DEG to the syrup, far exceeding the permitted limit of 0.1%.
Factory Violations and License Suspension
An inspection revealed unbilled containers of DEG in the Kancheepuram factory. Following these findings, the Tamil Nadu Drugs Control Authority issued a stop-production order, froze all stocks, and suspended the company’s license. Sresan Pharmaceuticals, originally registered in 1990, had continued operating even after being struck off the Ministry of Corporate Affairs register, raising concerns over regulatory oversight.
Widespread Impact and Bans
At least nine states have banned Coldrif syrup following the deaths. The case has exposed serious lapses in India’s pharmaceutical manufacturing and regulatory practices. The national drug regulator confirmed inspections showed not every batch of raw materials and active ingredients was being properly tested, highlighting systemic failures in ensuring medicine safety.









Achieve Radiant Skin at Home With This Simple Rice Flour Mixture
Say goodbye to costly facials! Discover how a rice flour concoction can rejuvenate your skin right f

Meta Unveils Paid Subscription Plans for Key Platforms
Meta introduces subscription services for Instagram, Facebook, and WhatsApp, offering users premium

2026 Eid Al Adha Dates Expected in UAE According to Astronomical Predictions
Astronomers anticipate Eid Al Adha in the UAE may start on May 27, 2026, prompting early holiday pla

DAE's First Quarter Financial Surge Sets New Highs
Dubai Aerospace Enterprise sees record first-quarter revenue and profit growth, alongside a major ac

Sony's PS5 Price Increase Set for Southeast Asia on May 1
Starting May 1, 2026, Sony will raise PS5 prices across Southeast Asia. Discover what this means for

Potential Super El Niño 2026: Understanding Climate Threats
Is a Super El Niño on the horizon for 2026? Explore its potential effects and global climate implica